Solar autonomy, This is our expertise!
We study, design & deploy autonomous solar systems with batteries reliable & durable, for all types of scenarios (isolated site, residential, tertiary & industry).
Solar autonomy systems for Isolated sites, Shelters, Tiny house, Serviced Holiday Lets Businesses Industries, Shops,



















Solar systems for isolated sites.
Robust & repairable.
Components from the most reliable brands (Studer, Victron, SMA).
Up to 10 kVA power (single phase).
SUN.CONNECT
Forget solar kits!
With SUNCONNECT, you get a tailor-made autonomous solar system, pre-wired and tested, guaranteeing simplified and secure installation.
Complete systems from 6990 € , out of pose.
What are the advantages ?
Unlike all autonomous solar kits sold on the internet, our pre-wired autonomous solar systems guarantee you a professional result, an installation that meets standards, and a secure system. They are ready to use and are pre-programmed and tested by us before shipping.
You choose the components used (Victron, Studer, SMA). If you want 100% European, it's possible!
Available capacities & powers
The powers of SUNCONNECT autonomous solar systems are available in:
3, 5, 8 & 10 kVA, single phase.
9 kVA three-phase (in cabinets).
They will be able to exploit up to 11 kWp of solar panels.
What possible uses?
Isolated sites (off-grid)
Hybrid sites (with EDF), for solar self-sufficiency
Batteries required.
Residential solar systems.
Simple to install, safe.
Scalable (with or without batteries).
100% functional in the event of an EDF outage.
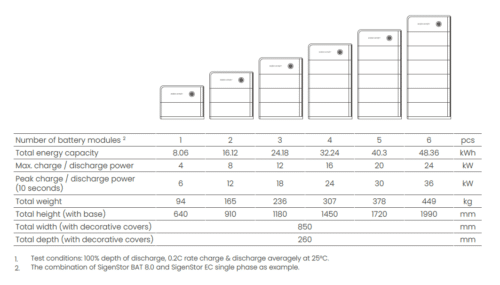
SIGENSTOR
High-performance solar station.
SIGENERGY revolutionizes solar autonomy with SIGENSTOR, an all-in-one solar station combining hybrid inverter and batteries, installable outdoors and modular over time.
Complete systems from 5000 € , out of pose.
The “tower” type format allows for minimal floor space, or wall mounting. Its installation is ultra-simplified thanks to its “interlocking” design of the elements. Future addition of batteries is possible. The system is IP66 and can be installed in front of a garage, and an EV charging module up to 22 kW DC can be added. The energy management modes are sophisticated and optimized thanks to AI. A very advanced smartphone application allows you to monitor the system in real time.
The SIGENERGY solar autonomy system is available in single and three-phase, from 3 to 25 kVA per turn.
The battery capacity starts at 5 kWh per revolution, and increases up to 48 kWh.
- Hybrid, with solar panels & EDF
- Back-up only, batteries without panels
- Solar panels alone, adding batteries later.
- V2H/V2L compatible possible.
- Off-grid.
Customized sizing for your needs.
Analysis of your consumption via LINKY data (optional)
Profitability analysis, estimation of your potential savings.
Design & study of your project.
Learn more about solar autonomy
Our range is intended for self-construction, so this requires a minimum of skills (installing your own solar panels, etc.). You can also choose to subcontract the installation to one of our partner installers, depending on your region. For tertiary battery projects, we have specially trained technicians who work throughout France.
The best battery will be the one that meets your objectives for your solar autonomy project. There are different criteria: cost, lifespan, size, power, and capacity. All these factors must fit best into your project thanks to the sizing that we will do with you. Nickel-Iron, Lithium LFP or LTO batteries each have advantages and disadvantages that must be taken into account.
Yes, all our solar systems are designed with modularity which will allow you to add solar panels and/or battery modules (only with lithium batteries).
Everything will depend on the energy context of the project. A home connected to EDF consuming 2 kWh per day will indeed have difficulty paying off solar batteries with a hybrid system at €9000. Conversely, on larger types of electricity consumption, a villa consuming 20 or 30 kWh for example, the return on investment (ROI) can be achieved over around ten years. This is without taking into account future electricity increases to come. For commercial or industrial buildings, with unregulated rates, profitability is even better, sometimes with ROI over 6 years! here is a example of profitability study in PDF format.
A solar self-sufficiency system is capable of operating in two schemes: in the presence of the EDF network (hybrid mode) OR on an isolated site (islanding), and in any way. In other words, they adapt to different scenarios and allow you to achieve energy autonomy. Due to their robustness, they can operate without EDF input without time limit, unlike certain systems (Fronius GEN24 for example).
We have selected a specific range of stand-alone solar systems, each with its own characteristics and benefits. This makes it possible to respond to each project, according to the specifications that you establish for us (budget, configuration, energy needs). For example, a SIGENERGY system will be fully integrated, simple to implement and modular, but it will not be suitable for people wishing to equip themselves with Nickel-Iron batteries, as the system is not compatible. In this case, you will have to opt for the SUNCONNECT range. Conversely, if you need a fully integrated modular system, the SIGENSTOR solution will be ideal.
Contact us!
For your tailor-made autonomous solar system project, consult us and obtain a free study and personalized quote.
We ship everywhere in France & Europe

















